5 Reasons Why You Should Perform Live Cell Imaging
ibidi Blog | October 4, 2022 | Tina Freisinger, ibidi GmbH
Cell biology is a discipline of the life sciences. As the name implies it is dedicated to investigating living beings. This fact alone should be reason enough to study cells in their natural, living state. But of course, there are additional profound reasons for live cell imaging. In this article, we name five good reasons why it makes sense to study living cells with time-lapse microscopy.
Background
Live cell imaging allows the in vivo observation of cells under the microscope over a certain period. Various microscopy techniques are suitable for live cell imaging. For example, one can either employ label-free techniques such as phase contrast, DIC, or interferometry or rely on fluorescence microscopy, which allows one to tag and visualize cellular substructures, molecules, or proteins with a fluorescent marker.
Of course, live cell imaging comes with challenges, and certain requirements are to be considered when setting up a live cell imaging experiment. Most importantly, one has to ensure that the microscope is equipped with a stage top incubator that can provide the ideal environment to keep the cells alive and healthy over time.
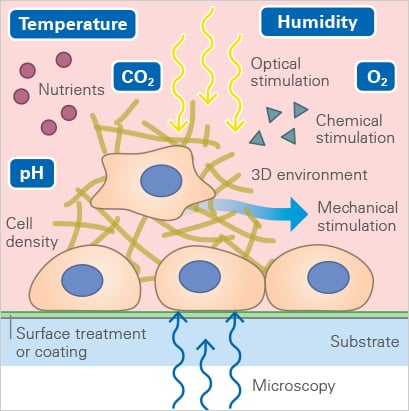
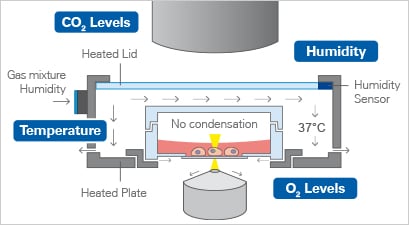
A
| B
Fig. 1A Environmental Parameters that need to be considered and controlled during live cell imaging. B: Schematic depiction of a stage top incubator for an inverted microscope |
The parameters and environmental conditions are an essential part of the story, which we will discuss in a future article.
In the meantime, if you are curious, read more about it here.
Now that we have covered the basics, we can dive right into the reasons why you should investigate your cells with live cell imaging:
1. Avoiding Artefacts from the Fixation Procedure
Cells are often fixed before (e.g., immunofluorescence) microscopy to preserve them in a lifelike state. Over the years, many different chemical and physical procedures have been optimized and established to maintain the quality of the original sample.
Nevertheless, the fixation process takes its toll on the cell (well, they are DEAD after this procedure) and irreversibly alters its organization, structure, and morphology (organelle shrinkage, protein mislocalization, etc.). In contrast, live cell imaging allows us to study cells that are, well, ALIVE. This means they should be displaying their natural behavior, which can still be affected by fluorescent tags, laser light, etc., but just like the environmental conditions, this is a story for a different time.
2. Observing and Analyzing Dynamic Processes
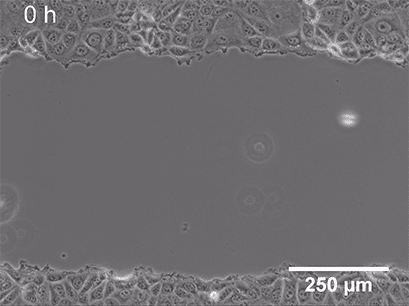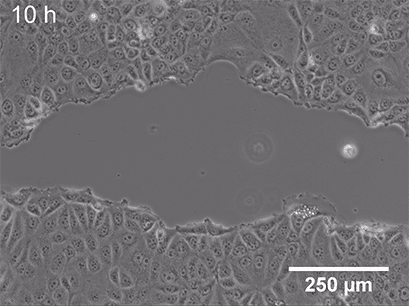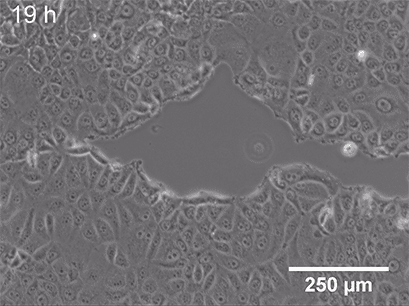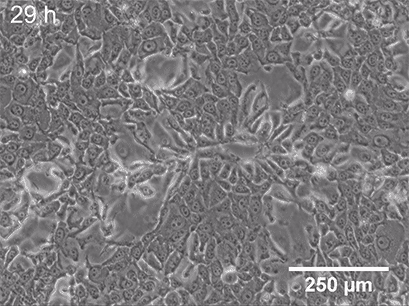
Live cell imaging allows us to observe dynamic events of a whole cell population, single cell, or even on a subcellular level. While fixing the cells locks them at a certain point in time in a specific (behavioral or structural) state, microscopy of living cells can give insights into a whole dynamic process. Functional cell-based assays such as wounding and migration (Fig 2) or chemotaxis experiments are great examples of live-cell imaging applications. These assays allow the investigation of cellular behavior in response to chemical (chemotaxis) or mechanical (wound healing) stimuli.
|
Fig. 2 Live cell imaging using the ibidi Stage Top Incubation System shows the gap closure of MCF7 cells in a wound healing and migration assay. Phase contrast; 10x objective lens. |
Find out more about functional cell-based assays here.
3. Following Cellular Changes in Real Time
Live cell microscopy is a valuable method to gain insights into spatiotemporal cellular changes in real time rather than relying on the results of endpoint analyses of fixed cells.
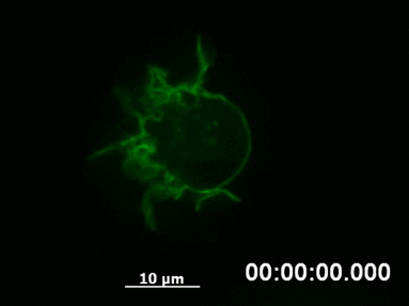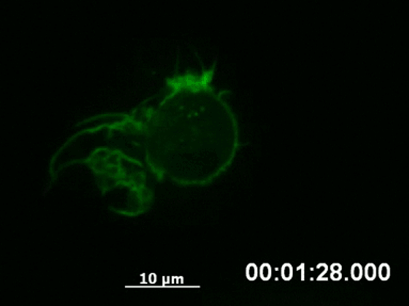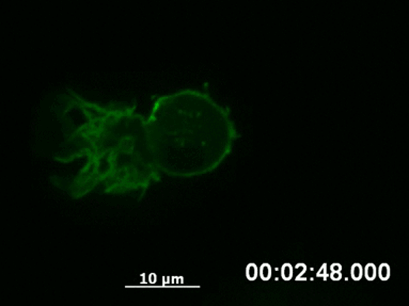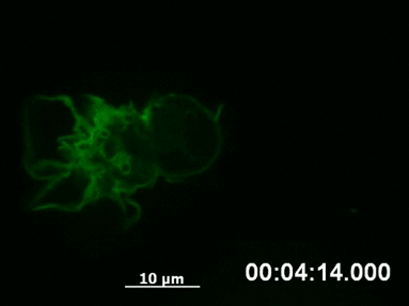
By following a cell over a longer period using time-lapse video microscopy, it is possible to capture the dynamics of structural rearrangements (e.g., polarization of the cytoskeleton after sensing a chemotactic stimulus, Fig. 3) or transient cellular events that would be missed using fixed cells (e.g., chromosome segregation during mitosis).
|
Fig. 3 Live cell imaging of actin dynamics in a LifeAct-expressing primary dendritic mouse cell after applying a chemotactic gradient. |
4. Investigating Single Molecule Dynamics, Localization, and Interaction
The development of advanced fluorescent labeling and imaging techniques, such as fluorescence recovery after photobleaching (FRAP), fluorescence lifetime imaging microscopy (FLIM), and fluorescence resonance energy transfer (FRET), have made the observation and analysis of single molecule localization, dynamics and interaction during live-cell imaging possible.
FRAP allows measurements of the mobility of fluorescently labeled molecules and proteins within living cells. FLIM provides information on the distribution of cellular molecules and their environment by measuring the lifetime of the attached fluorophores.
With FRET, one can measure the direct interaction of two molecules in living cells by detecting the interaction of their attached fluorophores when they come in nanometer proximity to each other.
5. Getting More Information Out of One Experiment
Overall, if you perform live cell imaging, you can get much more information from a single experiment than from imaging fixed cells. This is because live cell imaging allows one to follow molecule kinetics and dynamics and provides a bigger, more comprehensive picture of your cellular process of interest.
The analysis of fixed samples often only provides a snapshot of a certain cellular event, while following the whole dynamic process allows one to measure more parameters and draw more differentiated conclusions from one single experiment.
If you are interested in learning more about live cell imaging, visit our Live Cell Imaging Application Site. It offers you an overview on
• How to Investigate Biological Processes in Real Time
• Parameters for Live Cell Imaging
• Requirement and Setup of a Live Cell Imaging Experiment
• Experimental Examples
 (3)
(3)  (0)
(0)