Chemotaxis
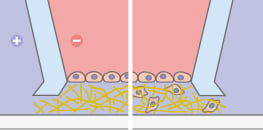
Chemotaxis refers to the directed movement of cells in response to chemical gradients, a fundamental process in various biological phenomena such as immune responses, wound healing, and cancer metastasis.
To study this behavior, chemotaxis assays are employed, allowing researchers to observe and quantify cell migration patterns under controlled conditions. These assays are crucial for analyzing cell motility, investigating disease mechanisms, and evaluating potential therapeutic agents.
ibidi facilitates these studies by offering specialized labware, such as the µ-Slide Chemotaxis, designed to create stable, reproducible gradients for high-resolution live cell imaging in both 2D and 3D environments. To learn more, explore the subchapters below, which cover key aspects of chemotaxis experiments, including gradient generation, analysis methods, and chamber system recommendations.

Explore the Chemotaxis Assay Guide
Follow the complete workflow from assay principle to experimental setup and quantitative data analysis.
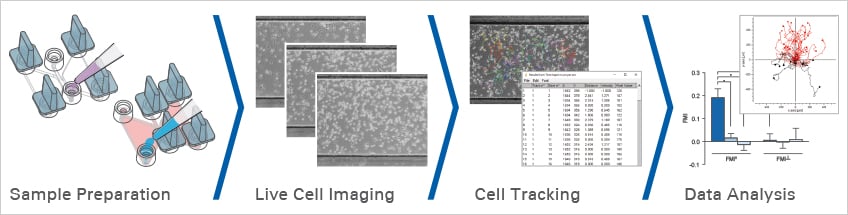
1. Chemotaxis Assay Principles
Understand chemical gradients, directed migration, and the differences between chemotaxis, chemokinesis, and random cell migration.
2. Chemotaxis Assay Workflow
Plan your experiment, prepare cells and matrices, generate gradients, and perform long-term live cell time-lapse imaging.
3. Chemotaxis Data Analysis
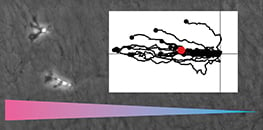
Track cells, visualize migration trajectories, and quantify parameters such as velocity, directionality, directness, and Forward Migration Index.
ibidi Products for Chemotaxis Assays
Find more products for chemotaxis assays.
Scientific Posters
Qualitative and quantitative analysis of slowly migrating chemotactic cancer cells in 3D (PDF)
Presented at the Experimental Biology (EB) Conference 2017, Chicago, Illinois, USA.
Presented at the Experimental Biology (EB) Conference 2018, San Diego, California, USA.
Selected Publications Using Chemotaxis Assays
Colorectal cancer cell chemotaxis was studied using the µ-Slide Chemotaxis.
Tetrick MG, Emon MAB, Doha U, et al. Decoupling chemical and mechanical signaling in colorectal cancer cell migration. Sci Rep. 2025;15(1):4952. 10.1038/s41598-025-89152-4.
Read article
Live cell imaging of hematopoietic stem and progenitor cells was performed in the µ-Slide Chemotaxis using the ibidi Stage Top Incubator.
Prchal-Murphy M, Zehenter J, Fischer M, et al. Repurposing the prostaglandin analogue treprostinil and the calcium-sensing receptor modulator cinacalcet to revive cord blood as an alternate source of hematopoietic stem and progenitor cells for transplantation. Front Pharmacol. 2025;15:1444311. 10.3389/fphar.2024.1444311.
Read article
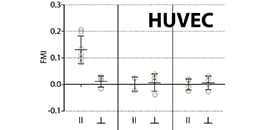
Live cell imaging of single HUVECs during chemotaxis was performed using the µ-Slide Chemotaxis.
Völkl M, Burgers LD, Zech TJ, et al. Homoharringtonine (omacetaxine mepesuccinate) limits the angiogenic capacity of endothelial cells and reorganises filamentous actin. Biomed Pharmacother. 2025;186:118025. 10.1016/j.biopha.2025.118025.
Read article
Chemotaxis of B16-F10 tumor cells was investigated using the µ-Slide Chemotaxis by generating a microenvironmental ROS gradient.
Ren X, Shi P, Su J, et al. Loss of Myo19 increases metastasis by enhancing microenvironmental ROS gradient and chemotaxis. EMBO Rep. 2024;25(3):971-990. 10.1038/s44319-023-00052-y.
Read article
The µ-Slide Chemotaxis was used to apply serum gradients across hydrogel clefts for studying 3D cell invasion.
Stöberl S, Balles M, Kellerer T, Rädler JO. Photolithographic microfabrication of hydrogel clefts for cell invasion studies. Lab Chip. 2023;23(7):1886-1895. 10.1039/d2lc01105k.
Read article
A combined 2D/3D chemotaxis assay for slow-moving cells was developed using the µ-Slide Chemotaxis.
Tomasova L, Guttenberg Z, Hoffmann B, Merkel R. Advanced 2D/3D cell migration assay for faster evaluation of chemotaxis of slow-moving cells. PLoS One. 2019;14(7):e0219708. 10.1371/journal.pone.0219708.
Read article
EGF-guided chemotaxis of MDA-MB-231 breast cancer cells was analyzed using the µ-Slide Chemotaxis.
Biswenger V, Baumann N, Jürschick J, Häckl M, Battle C, Schwarz J, Horn E, Zantl R. Characterization of EGF-guided MDA-MB-231 cell chemotaxis in vitro using a physiological and highly sensitive assay system. PLoS One. 2018;13(9):e0203040. 10.1371/journal.pone.0203040.
Read article
Application Notes for Chemotaxis Assays
Chemotaxis Assay Videos
User Protocols for Chemotaxis Assays
Frequently Asked Questions
What is chemotaxis and why is it studied?
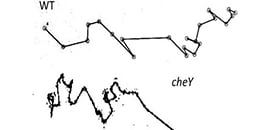
Chemotaxis is the directed movement of cells along a chemical gradient. It plays a key role in immune response, tissue repair, angiogenesis, and cancer metastasis. Studying chemotaxis helps to distinguish directional guidance from general motility and reveals signaling pathways that control cell migration.
How is chemotaxis different from chemokinesis?
Chemotaxis describes directional migration along a gradient, whereas chemokinesis refers to changes in migration speed or activity without a preferred direction. Proper controls are required to distinguish between both effects in a chemotaxis assay.
What is a gradient in a chemotaxis assay?
A gradient is the spatial change in concentration of a chemoattractant. It represents the slope of a concentration profile and determines the direction of cell migration. Cells respond to concentration differences rather than absolute concentrations, making controlled gradient formation essential for reproducible chemotaxis experiments.
How are stable gradients generated in chemotaxis assays?
In diffusion-based systems, two reservoirs with different concentrations are connected by an observation area where a gradient forms. Systems such as the µ-Slide Chemotaxis enable rapid gradient formation and maintains stable conditions for more than 48 hours, supporting reliable long-term live cell imaging.
Why are live cell imaging and cell tracking required for chemotaxis assays?
Chemotaxis assays are not endpoint assays. Since most cells migrate with low directionality, differences between start and endpoint cannot be reliably detected by simple counting. Time-lapse imaging and single-cell tracking are required to quantify migration direction, speed, and trajectory.
Which controls are needed in a chemotaxis assay?
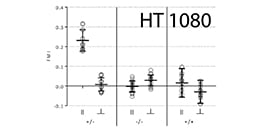
A chemotaxis assay should include a negative control without chemoattractant (-/-), a positive control with equal chemoattractant concentration on both sides (+/+), and a gradient condition (+/-). These controls help distinguish directed chemotaxis from random migration and chemokinesis.
When should I use a 2D or a 3D chemotaxis assay?
2D assays are easier to handle and ideal for standardized experiments and screening. 3D assays embed cells in matrices such as collagen or Matrigel, providing a more in vivo-like environment. The choice depends on the biological question and the physiological relevance required.
How is chemotaxis quantified?
Cells are tracked over time to generate trajectories. From these, parameters such as Forward Migration Index, center of mass, directness, and speed are calculated. Statistical methods such as the Rayleigh test are used to determine whether migration is directional.
Which ibidi products support chemotaxis workflows?
The µ-Slide Chemotaxis enables reproducible gradient formation for 2D and 3D assays. Additional tools such as Stage Top Incubators, Collagen Type I, and the Chemotaxis and Migration Tool support the complete workflow from sample preparation to quantitative data analysis.
List of pages in %s: