Neural 3D Cell Models
Neural 3D cell culture is a technique used to grow and study neural cells in a three-dimensional environment that mimics the complex structure and function of the brain and peripheral nervous system. In traditional 2D cell culture, cells are grown on a flat surface, but this approach does not capture the complex interactions between the cells and their surrounding tissue.
3D neural cell culture has a wide range of applications, including the study of neural development (e.g., the generation of neurospheres from neural stem cells), disease modeling, drug discovery, and neural tissue engineering techniques.
In 3D cell culture, cells are embedded in a three-dimensional matrix made of materials such as hydrogels (e.g., collagen), extracellular matrix proteins, or synthetic polymers. This matrix provides a scaffold for the cells to grow and form complex structures, such as neural networks, spheroids, or organoids, that more closely resemble the tissue architecture of the nervous system.
A 3D environment allows cells to interact with each other in a way that more closely mimics the complex interactions that occur in vivo, leading to more physiologically relevant results. Furthermore, 3D cell culturing techniques lead to improved cell viability and differentiation: Cells grown in 3D matrices have an improved ability to maintain their viability and differentiate into specific cell types compared to cells grown in 2D.

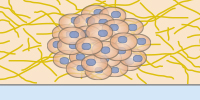
Neurosphere formation after retinoic acid-induced differentiation of murine neural stem cells (NE-4C) on poly-L-lysine-coated glass. Neurons were stained with Tuj1 (green), astrocytes with GFAP (red), and nuclei with DAPI (blue). The image was acquired with a 20x objective on a Leica Microsystems SP8 confocal microscope.
Image courtesy: Lina Papadimitriou, Anthi Ranella, TERMIM Lab, Institute of Electronic Structure and Laser, FORTH, Crete, Greece
Find Out MorePlease find more detailed information about 3D cell culture assays here. |
|
ibidi Solutions for Neural 3D Models
The ibidi Collagen Type I (bovine or rat tail origin) is a non-pepsinized, native collagen for modeling ECM in gel matrices. Its fast polymerization facilitates optimal cell distribution in 3D gels. Read our Application Note about how to prepare a 3D gel using the ibidi Collagen Type I here: AN 26: Collagen I Gel for 3D Cell Culture. |
|
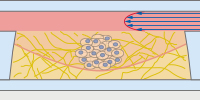
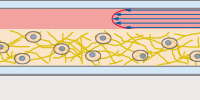
The µ-Slide Spheroid Perfusion is a specialized flow chamber for long-term spheroid culture. Each of the 3 x 7 wells forms its own niche, in which the specimen is cultured. The application of perfusion through the channel on top of the wells (e.g., by using the ibidi Pump System) ensures optimal nutrition and oxygen diffusion throughout the experiment without exposing the specimen to significant shear forces. |
|
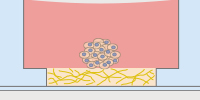
The µ-Slides with Multi-Cell µ-Pattern enable spatially defined cell adhesion for spheroid and organoid generation, long-term culture, and high resolution imaging. Defined adhesion spots are able to catch all adherent single cells from a cell suspension. The surrounding Bioinert surface is fully non-cell-attachable. This forces all cells to aggregate to each other at the adhesion spots, thus forming spheroids in a defined and controllable way |
|
Bioinert is a stable, biologically inert surface for long-term culture and high-resolution microscopy of spheroids, organoids, and suspension cells on a non-adherent surface without any cell or biomolecule adhesion. It is currently available as the µ-Dish 35 mm, high Bioinert, the µ-Slide 8 Well high Bioinert, the µ-Slide 4 Well Bioinert, the µ-Slide VI 0.4 Bioinert, and the µ-Slide Spheroid Perfusion Bioinert. |
|
In the µ-Slide III 3D Perfusion, single cells, spheroids, or organoids can be cultivated in or on a gel layer or embedded in a 3D matrix. The special channel geometry allows for perfusion with a low flow rate (e.g., when utilizing the ibidi Pump System), ensuring optimal oxygen and nutrient supply. This setup makes long-term cultivation possible for up to several weeks. Additionally, the thin coverslip bottom allows for high-resolution imaging. |
|
The µ-Slide 15 Well 3D and the the µ-Plate 96 Well 3D are easy, cost-effective solutions for the 3D cultivation and microscopy of single cells, co-cultures, spheroids, and organoids on or in gel matrices. The gel layer is directly connected to the medium reservoir above, which enables fast and easy medium exchange by diffusion. The µ-Slide 15 Well 3D Glass Bottom, with a No. 1.5H glass bottom, is also available for special applications. |
|
The μ-Slide I Luer 3D is designed for culturing cells on or in a 3D gel matrix with defined shear stress. Each of the three wells can be filled with a gel, in which cells can be embedded. For defined flow application, the channel on top can be connected to a pump (e.g., to the ibidi Pump System) to ensure optimal oxygen and nutrient supply. |
|
Selected References
Imaging of 3D Neuronal Co-Cultures
The ibidi µ-Slide 8 Well was used to image 3D cultures of neurons and meningeal fibroblasts in hydrogels.
Santos TE, Schaffran B, Broguière N, Meyn L, Zenobi-Wong M, Bradke F. Axon Growth of CNS Neurons in Three Dimensions Is Amoeboid and Independent of Adhesions. Cell Rep. 2020 Jul 21;32(3):107907. doi: 10.1016/j.celrep.2020.107907.
Read article
Imaging of Humanized Three-Dimensional Peripheral Neurovascular (PNV) Models
The µ-Slide 8 Well was used to image a 3D co-culture model comprised of human embryonic stem cell peripheral sensory neurons (hESC)(PSNs) and hESC endothelial cells mixed with collagen type 1.
Kannan S, Lee M, Muthusamy S, Blasiak A, Sriram G, Cao T. Peripheral sensory neurons promote angiogenesis in neurovascular models derived from hESCs. Stem Cell Res. 2021 Apr;52:102231. doi: 10.1016/j.scr.2021.102231.
Read article
Read on and learn more about Neurological Conditions, Disorders, and Diseases, Neural Cell Analysis, and Neural Function and Behavior.