3D Models of Cancer
Tumor cells grow in a three-dimensional microenvironment, where they communicate and interact with each other and their surroundings. When cultured inside a 3D matrix, cells behave differently compared to a 2D environment. In many cases, a 3D cell culture setup more accurately reflects the in vivo situation. This includes drug screenings that use spheroids and organoids, which are indispensable nowadays as tumor models. This should be considered when analyzing cancer cell behavior, migration, proliferation, response to drug treatment, and gene and protein expression.
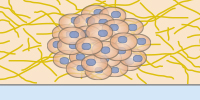
Invasion of HT-1080 cancer cells in a 3D Collagen gel. Invasive human fibrosarcoma cancer spheroids (HT-1080) were embedded into Collagen Type I, Rat Tail gel. The invasion into the gel matrix was recorded for 48 hours in the µ-Slide 8 Well. 4x objective lens, brightfield.
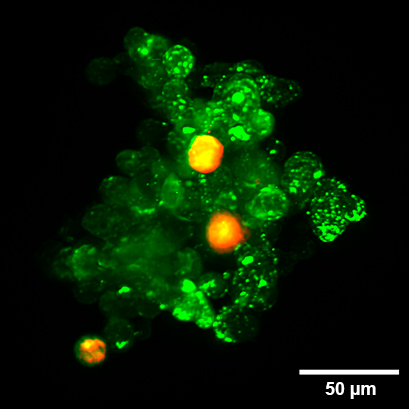
Organoid co-culture of PDAC cells and Fibroblasts. Organoid co-culture of the human pancreatic cancer (PDAC) cell line PA-TU-8988T (green, stained with CellTrackerTM Green) and the murine fibroblast cell line mPSC4 (red, stained with CellTrackerTM Orange CMTMR) in the µ-Slide Spheroid Perfusion. The µ-Slide was covered with a 25 µm FEP foil for matching the refractive index closer to water during upright light sheet microscopy. The image was acquired by S. Volkery at MPI Muenster with the M Squared Aurora Airy beam upright light sheet setup. The sample was provided by K. Roth, University Marburg, Germany.
ibidi Solutions for 3D Cancer Models
The ibidi Collagens Type I are non-pepsinized, native collagens for modeling ECM in gel matrices. Their fast gelation facilitates optimal cell distribution in 3D gels. Read our Application Note about how to prepare a 3D gel using the ibidi Collagen Type I here: AN 26: Preparation of Collagen I Gels. |
|
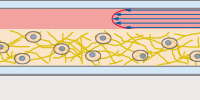
The µ-Slide Spheroid Perfusion is a specialized flow chamber for long-term spheroid culture. Each of the 3 x 7 wells forms its own niche, in which the specimen is cultured. The application of perfusion through the channel on top of the wells (e.g., by using the ibidi Pump System) ensures optimal nutrition and oxygen diffusion throughout the experiment, without exposing the specimen to significant shear forces. |
|
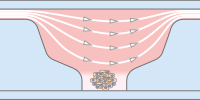
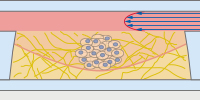
The ibidi µ-Pattern ibiTreat with multi-cell patterns enable spatially defined cell adhesion for spheroid and organoid generation, long-term culture, and high resolution imaging. Defined adhesion spots are able to catch all adherent single cells from a cell suspension. The surrounding Bioinert surface is fully non-cell-attachable. This forces all cells to aggregate to each other at the adhesion spots, thus forming spheroids in a defined and controllable way. |
|
Bioinert is a stable, biologically inert surface for long-term culture and high-resolution microscopy of spheroids, organoids, and suspension cells on a non-adherent surface without any cell or biomolecule adhesion. It is currently available as the µ-Dish 35 mm, high Bioinert, the µ-Slide 8 Well high Bioinert, the µ-Slide 4 Well Bioinert, and the µ-Slide VI 0.4 Bioinert. |
|
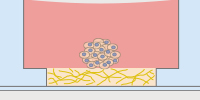
In the µ-Slide III 3D Perfusion, single cells, spheroids, or organoids can be cultivated in or on a gel layer or embedded in a 3D matrix. The special channel geometry allows for superfusion with a low flow rate (e.g., when utilizing the ibidi Pump System), ensuring optimal oxygen and nutrient supply. This setup makes long-term cultivation possible for up to several weeks. Additionally, the thin coverslip bottom allows for high-resolution imaging. |
|
The µ-Slide 15 Well 3D and the µ-Plate 96 Well 3D are easy, cost-effective solutions for the 3D cultivation and microscopy of single cells, co-cultures, spheroids, and organoids on, or in, gel matrices. The gel layer is directly connected to the medium reservoir above, which enables fast and easy medium exchange by diffusion. For special applications, the µ-Slide 15 Well 3D Glass Bottom, with a No. 1.5H glass bottom, is also available. |
|
The μ-Slide I Luer 3D is designed for culturing cells on, or in, a 3D gel matrix with defined shear stress. Each of the three wells can be filled with a gel, in which cells can be embedded. For defined flow application, the channel on top can be connected to a pump (e.g., to the ibidi Pump System) to ensure optimal oxygen and nutrient supply. |
|
Selected References
3D Sandwich culture of squamous cell carcinoma lines using the µ-Plate 96 Well 3D
Hoque Apu, E., Akram, S. U., Rissanen, J., Wan, H., & Salo, T. (2018). Desmoglein 3 – Influence on oral carcinoma cell migration and invasion. Experimental Cell Research. 10.1016/J.YEXCR.2018.06.037
Read article
Breast tumor organoid culture in the µ-Slide 15 Well 3D
Lüönd, F., Sugiyama, N., Bill, R., Bornes, L., Hager, C., Tang, F., Santacroce, N., Beisel, C., Ivanek, R., Bürglin, T., Tiede, S., van Rheenen, J., & Christofori, G. (2021). Distinct contributions of partial and full EMT to breast cancer malignancy. Developmental Cell. 10.1016/J.DEVCEL.2021.11.006
Read article
Live cell imaging of HT29 tumor spheroid co-culture with neutrophils and NET formation in the µ-Slide III 3D Perfusion
Teijeira, Á., Garasa, S., Gato, M., Alfaro, C., Migueliz, I., Cirella, A., de Andrea, C., Ochoa, M. C., Otano, I., Etxeberria, I., Andueza, M. P., Nieto, C. P., Resano, L., Azpilikueta, A., Allegretti, M., de Pizzol, M., Ponz-Sarvisé, M., Rouzaut, A., Sanmamed, M. F., … Melero, I. (2020). CXCR1 and CXCR2 Chemokine Receptor Agonists Produced by Tumors Induce Neutrophil Extracellular Traps that Interfere with Immune Cytotoxicity. Immunity. 10.1016/J.IMMUNI.2020.03.001
Read article