Cell Culture: Sample Preparation for Imaging
A well-planned experimental setup, as well as the optimized, reproducible handling of cells before imaging, are the keys for a successful microscopy experiment. To this end, several factors must be considered, and these depend on the microscopy method and the general setup being used:

What are the crucial questions for planning an experiment?
Are you choosing the optimal vessel geometry for your experiments and microscopy technique?
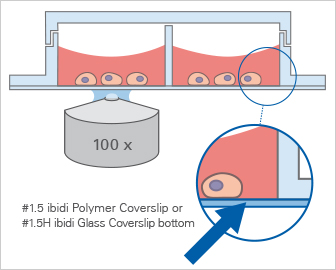
A large variety of cell culture vessels exist to meet the complexity of diverse cell culture assays, such as experiments using suspension or adherent cells, 2D or 3D assays, single or co-culture assays, and many more. The main culture vessel geometries (e.g., growth area and volume) are important factors that affect the outcome and interpretation of an experiment, and should be carefully considered before starting. Additionally, the vessel geometries influence the physical (e.g., evaporation) and optical (e.g., meniscus effect) properties in the experiments.
ibidi SolutionsWe provide imaging chambers with various geometries that are specifically tailored to your assay. Get more detailed information in our application chapter, “The Geometry of the ibidi Chambers”. |
Did you choose a suitable substrate, surface, and coating for your specific cell type and microscopy method?
The growth, development, and signaling of cultured cells strongly depends on the surface being used for cell seeding. Additionally, the optical specifications strongly determine which substrate is advisable. Borosilicate glass is widely used as a substrate, however, a coating might be necessary to support cell adherence. The use of polymers is also very popular. They are usually tissue culture-treated to allow for direct cell adherence; however, not all polymers available on the market are suitable for microscopy. Labware with the ibidi Polymer Coverslip bottom provides the highest optical quality for various high-resolution microscopy applications.
However, if the cells require a specific extracellular matrix (ECM) on the surface, then coatings such as collagen I, collagen IV, or poly-L-lysine can be used. For suspension cells or spheroid culture, a non-adherent surface, such as ibidi Bioinert, should be used.
Also, it should be carefully considered whether a 2D or a 3D experiment will be performed. For more details, read our extensive Application Chapter about 3D Cell Culture Assays.
ibidi Solutionsibidi Labware |
Collagen Type I, Rat Tail |
|
Non-Adherent Bioinert Surface |
|
Micropatterning |
|
Do you continuously take care of sterile and reproducible cell handling?
For all experiments, it is crucial to keep the conditions sterile, homogeneous and reproducible, in order to get comparable results.
Read on and learn more details about Cell Handling and Prevention of Contaminations.
Are you ensuring that the cells are viable and healthy when they are being cultured, as well as throughout the whole imaging procedure?
Microscopy can be done with both living and fixed cells, depending on the experimental setup and the required readout. In living cells, the microscopy procedure causes stress. The cells have to be taken out of the incubator, which leads to a reduction in the surrounding temperature and humidity. Additionally, the cells are exposed to light for a certain amount of time, which might cause phototoxicity. Generally, all microscopy steps should be carried out as quick as possible—while maintaining precision—to minimize cell stress.
Therefore, only viable and healthy cells should be used for any imaging experiment. To ensure this, cell attachment, morphology, confluency, nutrient supply, and signs of contamination should be controlled daily under the microscope. Plus, several parameters, such as temperature and humidity, must be kept consistent.
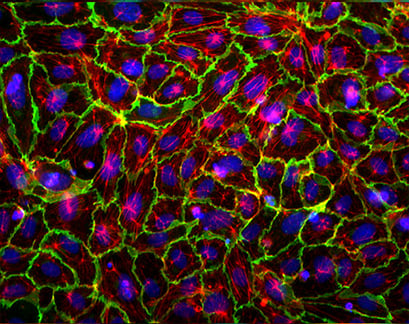
Immunofluorescence of Human Umbilical Vein Endothelial Cells (HUVEC) in a µ-Slide 4 Well
Read more about how to prevent phototoxicity in our application chapter, "Image Acquisition", or learn more about Cell Handling or Parameters for Healthy Cells.
Can you ensure that the optical requirements for your microscopy technique of choice are being met?
Every microscopy technique has its own special requirements for the substrate and cell pretreatment, and these should be considered when planning the experiment. You can get more detailed information about the optical parameters of the different substrates in our application chapters, "Microscopy Parameters of Materials" and "Comparison of Material Specifications".
Furthermore, some general recommendations apply before starting microscopy:
- All media should be equilibrated in order to prevent the formation of air bubbles inside the vessels. By minimizing temperature differences, external convective currents can be avoided that can considerably disturb microscopy.
- When performing fluorescence microscopy, it is especially crucial to minimize cell stress caused by excitation light, because this might alter the outcome of an experiment. Please get more detailed insights about provisions against phototoxicity here.
- Also, autofluorescence of cell culture vessels or media can be an unwanted side effect. Therefore, specialized cell culture vessels with low autofluorescence should be applied for fluorescence microscopy.
ibidi Solutions
Many microscopy techniques, such as phase contrast, widefield fluorescence, and confocal microscopy, are possible without restrictions when using any of the ibidi labware that contain the ibidi Polymer Coverslip or the ibidi Glass Coverslip bottom. |  |
When planning a live cell imaging experiment, does your setup meet all the necessary requirements?
If several images in a row, or at specific time points, are needed, then live cell imaging might be the best choice for you. To keep the cells alive and healthy during a live cell imaging experiment, physiological conditions must be established and maintained on the microscope—the same set up as inside the incubator.
Get more information on how to plan and perform a live cell imaging experiment in our application chapter here.

ibidi Solution
The ibidi Stage Top Incubation Systems are available for slides/dishes and for multiwell plates. They allow for the accurate and reliable control of important live cell imaging parameters (temperature, CO2 and O2 concentration, and relative humidity) during short-term and long-term assays on any inverted microscope. If an additional control of the oxygen level is needed (e.g., in hypoxia experiments), the ibidi Stage Top Incubation Systems, CO2/O2 are the ideal solution. |
|
Read on and learn more about Handling Cells in Culture.