Epithelial Cell Barrier Function
Impedance Measurements
Measuring barrier function (i.e., the resistance of the paracellular pathway between cells) has proven to be a significant and rapidly growing application in ECIS technology.
The use of ECIS technology to monitor barrier function was first demonstrated in 1992 in an experiment using bovine pulmonary endothelial cells exposed to thrombin. Recent data has suggested that various cellular effects can be monitored at different frequencies used during experimentation. While cell spreading can be analyzed at high frequencies (analyzing capacitance data), the formation or disruption of a barrier function is best described at low frequencies (analyzing impedance data).
For more information on this subject, please refer to: J. Wegener. Impedance Analysis of Cell Junctions. Nanotechnology Ed. H. Fuchs. VCH Weinheim, 2009.
The use of ECIS technology to monitor barrier function was first demonstrated in 1992 in an experiment using bovine pulmonary endothelial cells exposed to thrombin:
C. Tiruppathi, A.B. Malik, P.J. Del Vecchio, C.R. Keese and I. Giaever. Electrical method for detection of endothelial cell shape change in real time: assessment of endothelial barrier function. PNAS, 1992, 10.1073/pnas.89.17.7919.
Read article
Experimental Example: Effect of Barrier Disruption on Epithelial Cells
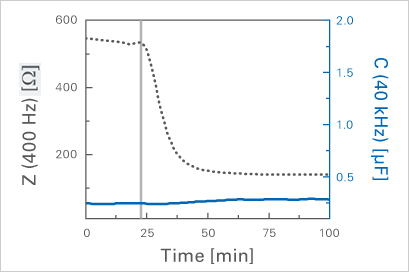
The capacitance (C) of the cell layer covering the electrode remains constant after disruption of the cell-cell contacts while impedance (Z) breaks down dramatically.

Staining of ZO-1 protein in epithelial cells (MDCK) showing barrier disruption after addition of Cytochalasin D (CD). The fungal toxin CD is known to depolymerize the intracellular actin filaments and to open the paracellular contacts between the cells, while the cell membrane is still intact.
Appropriate electrode arrays could be:
|
8 well format: |
96 well format: |

Optical Measurements
Optical measurements of barrier function are possible with all ibidi μ-Slides and μ-Dishes when used in conjunction with fluorescent dyes.
Signal Transduction
Impedance Measurements
Monitoring epithelial and endothelial barrier function plays a role during drug development (e.g., for drugs that change the paracellular permeability). G protein-coupled receptors (GPCR) are pharmacologically highly relevant, as their binding with a ligand (agonist) triggers an intracellular signaling cascade, leading to a transient or permanent barrier increase or decrease. Any GPCR activation, regardless of the second messenger, results in alterations of cytoskeletal elements and culminates in morphological changes of the cell. This event is precisely detected in real-time and with great sensitivity by the ECIS System.
For further information:
Stolwijk JA, Matrougui K, Renken CW, Trebak M. Impedance analysis of GPCR-mediated changes in endothelial barrier function: overview and fundamental considerations for stable and reproducible measurements. Pflugers Arch. 2015, 10.1007/s00424-014-1674-0.
Read article
Experimental Example
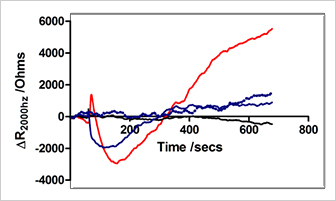
Changes in barrier function in human dermal microvascular endothelial cells (HDMECs) measured as resistance at 2000 Hz due to addition of:
serum free medium (black line),
2.5 µM Thapsigargin (red line),
12.5 nM Histamine (blue line),
and 12.5 nM Histamine after incubation in 2.5 uM Thapsigargin (dotted blue line).
The following electrode arrays are ideally suited for signal transduction studies:
|
8 well format: |
96 well format: |






