Wound Healing and Migration
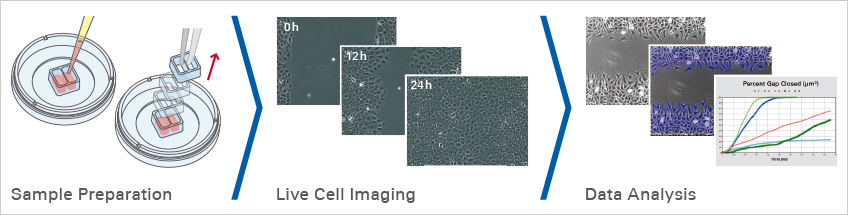
Wound healing assays and cell migration assays are widely used to study how cells move collectively across a surface. By creating a defined cell-free gap within a confluent monolayer and monitoring gap closure over time, researchers can quantify migration speed, directionality, and cell–cell interactions under controlled conditions. Cell migration is essential for embryonic development, tissue repair, inflammation, angiogenesis, and cancer metastasis. In vitro wound healing models allow researchers to investigate how genetic modifications, mechanical cues, or pharmacological treatments influence collective cell behavior in a reproducible 2D environment.
To ensure consistent and reliable results, ibidi offers dedicated tools such as the Culture-Insert 2 Well, Culture-Insert 3 Well, and Culture-Insert 4 Well, which generate highly defined 500 µm gaps without damaging the cell layer or substrate. These inserts can be combined with imaging-optimized vessels like the µ-Dish 35 mm, high or the µ-Plate 24 Well, and live monitoring can be performed using the Stage Top Incubation System under controlled temperature and CO2 conditions.
ibidi Blog Article |
From wound healing to placenta biology, Derek Sung (University of Pennsylvania) shares his research journey and how ibidi Culture-Inserts became a game-changer in his blog articles “Time Heals All Wounds” and “The Placenta: Here for a Good Time, Not a Long Time”.
To see how these themes connect to the broader role of cell migration in health and disease, check out the latest ibidi Blog article “The Great Escape: How Cells Migrate to Shape Life and Disease.”
Selected Publications for Wound Healing and Migration Assays
The Chamber Gap Assay was established based on the Culture-Insert 2 Well to enable reproducible and directional macrophage migration toward pancreatic cancer cells with live cell tracking.
Lenz M, Muliawan S, Nowak F, et al. The Chamber Gap Assay Is a Simple and Sensitive In Vitro Method for Studying Pancreatic Cancer-Induced Macrophage Recruitment and Morphological Alteration. Biology. 2026;15:240. doi:10.3390/biology15030240.
Read article
Wound closure and epithelial barrier repair were analyzed using the Culture-Insert 3 Well in a µ-Dish 35 mm, high to generate defined 500 µm cell-free gaps.
Lechuga S, Marino-Melendez A, Davis A, et al. Coactosin-like protein 1 regulates integrity and repair of model intestinal epithelial barriers via actin binding dependent and independent mechanisms. Front Cell Dev Biol. 2024;12:1405454. doi:10.3389/fcell.2024.1405454.
Read article
The Culture-Insert 2 Well was used to investigate how ITIH5 suppresses pancreatic cancer cell adhesion and migration in defined gap closure assays.
Kosinski J, Sechi A, Hain J, et al. ITIH5 as a multifaceted player in pancreatic cancer suppression, impairing tyrosine kinase signaling, cell adhesion and migration. Mol Oncol. 2024;18(6):1486-1509. doi:10.1002/1878-0261.13609.
Read article
Keratinocyte migration under hypoxic conditions was quantified using the Culture-Insert 4 Well in a µ-Dish 35 mm, high to create standardized 500 µm wound gaps.
Yan T, et al. Hypoxia Regulates mTORC1-Mediated Keratinocyte Motility and Migration via the AMPK Pathway. PLOS ONE. 2017;12:e0169155. doi:10.1371/journal.pone.0169155.
Read article
Find more publications in the ibidi Reference Database.
Application Notes for Wound Healing and Migration Assays
Webinars and Videos for Wound Healing and Migration Assays
Frequently Asked Questions
What is a wound healing assay and what does it measure?
A wound healing assay is a 2D cell migration assay in which a defined gap is created within a confluent cell monolayer. Over time, cells migrate into this gap, and the reduction of the cell-free area is measured as an indicator of migration. The primary readout is gap closure rate, but additional parameters such as migration speed, collective behavior, and cell morphology can also be analyzed.
How is a gap created in a wound healing assay?
A gap can be generated either mechanically by scratching the monolayer or by using a physical insert that prevents cell growth in a defined region. The ibidi Culture-Inserts create highly reproducible and clean 500 µm gaps without damaging the surface or the cells. After removal of the insert, two precisely defined cell fronts migrate toward each other, enabling consistent quantitative analysis.
What are the advantages of Culture-Inserts compared to a scratch assay?
In a traditional scratch assay, gap width varies depending on pressure and tip size, and the surface coating may be damaged. This can affect cell adhesion and migration behavior. In contrast, the ibidi Culture-Inserts generate defined gaps without mechanical stress or debris, improving reproducibility and reducing experimental variability.
Can wound healing assays be used for co-culture or invasion studies?
Yes. The Culture-Insert 3 Well and Culture-Insert 4 Well allow seeding of different cell types or treatments within the same vessel. This enables co-culture migration assays and 2D invasion experiments, where interactions between tumor cells, fibroblasts, or immune cells can be studied under identical culture conditions.
How is migration monitored during the experiment?
Migration can be analyzed by taking images at defined time points or by performing continuous live cell imaging. Using the Stage Top Incubation System, cells can be monitored under stable physiological conditions during long-term imaging. Phase contrast microscopy is commonly used, but fluorescence imaging is also possible when studying labeled cells.
Should cell proliferation be inhibited during a wound healing assay?
Cells not only migrate into the gap but may also proliferate. If the goal is to analyze migration independently of proliferation, mitotic inhibitors such as mitomycin C can be applied before gap creation. Appropriate controls are essential to ensure that the treatment does not affect migration behavior itself.
What factors are critical for reproducible wound healing assays?
Reproducibility depends on standardized cell culture conditions, consistent seeding density, uniform gap creation, and controlled imaging parameters. Using defined tools such as ibidi Culture-Inserts, imaging-optimized vessels like the µ-Dish 35 mm, high or the µ-Plate 24 Well, and stable environmental control systems helps minimize variability and ensures robust quantitative results.
List of pages in %s: