In Vitro Models of the Nervous System
ibidi Blog | April 29, 2026 | Abhishek Derle, ibidi GmbH
Understanding the nervous system is not just about studying cells. It is about decoding one of the most complex systems in biology. Neurons form dynamic networks, constantly communicating through electrical and chemical signals while interacting with astrocytes, microglia, and their surrounding environment. Recreating this complexity in the lab is challenging, but essential for studying development, function, and disease [1].
This is where in vitro models come in. They allow researchers to simplify complexity just enough to study specific mechanisms while still capturing key biological behaviors. Over time, these models have evolved from simple flat cultures to sophisticated 3D systems, organoids, and controlled microenvironments [2]. Each model tells a different part of the story.
Flat but Powerful: Why 2D Neuronal Cultures Still Matter
Two-dimensional neuronal cultures are often the starting point in neuroscience, and for good reason. They provide a clean, controlled environment where individual cells and their behavior can be observed with high precision. In 2D systems, neurons grow on flat surfaces, making it easy to track neurite outgrowth, polarization, and synapse formation, while providing unmatched optical accessibility for high resolution fluorescence imaging, calcium imaging, and live cell analysis [3].
This simplicity allows researchers to focus on specific cellular mechanisms while enabling highly quantitative analysis. Today, 2D neuronal cultures are central to automated imaging workflows, where thousands of cells are analyzed using high content imaging and AI-based analysis [4]. Imaging optimized formats such as the µ-Plate 96 Well and µ-Plate 384 Well support these approaches with consistent conditions and reliable optical quality, making it easier to detect subtle phenotypic changes.
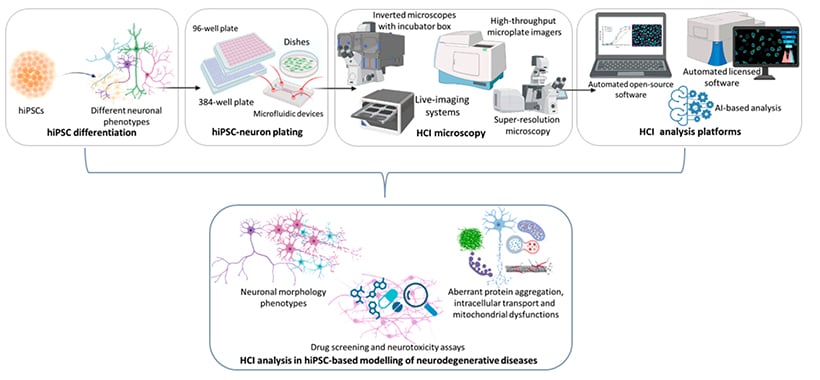
Fig. 1 | Workflow for high content imaging and analysis in hiPSC-based modeling of neurodegenerative diseases [3].
However, neurons in 2D still lack the spatial context and mechanical cues present in native tissue, which can influence how networks form, connect, and respond to stimuli. Still, 2D cultures remain an essential foundation, especially when precision and reproducibility are key.
Stepping into the Third Dimension
Moving from 2D to 3D does not just change the geometry, it changes how neurons behave. Instead of spreading on a flat surface, cells begin to navigate a surrounding matrix, making decisions about where to grow, connect, and stabilize their networks. This three-dimensional environment introduces spatial constraints, local gradients, and mechanical resistance, all of which influence how neuronal networks form and evolve over time.
This creates a more realistic microenvironment where cells interact not only with each other but also with the surrounding matrix, resulting in more complex networks, improved differentiation, and more physiologically relevant behavior. Neural spheroids are a common example of cells self-organizing into dense, interconnected structures that mimic aspects of tissue organization.
Within 3D culture systems, brain organoids represent a higher level of biological complexity. Derived from pluripotent stem cells, including induced pluripotent stem cells, these systems self-organize into tissue-like architectures with multiple neural cell types and region-specific identities. These systems allow researchers to recreate key aspects of human brain development and function in vitro, from early patterning to network formation, and to investigate neurodevelopmental disorders, neurodegeneration, and viral infections affecting the brain [5, 6].
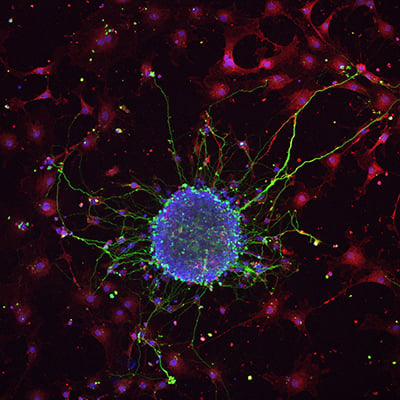
Fig. 2 | Neurosphere formation after retinoic acid-induced differentiation of murine neural stem cells on poly-L-lysine-coated glass. Neurons were stained with Tuj1, astrocytes with GFAP, and nuclei with DAPI. Image courtesy: Lina Papadimitriou, Anthi Ranella, TERMIM Lab, Institute of Electronic Structure and Laser, Crete, Greece.
What makes modern 3D systems especially powerful is the ability to combine structural complexity with controlled environments. Defined formats such as the µ-Plate 96 Well 3D enable reproducible gel-based assays in a high throughput setting, while the µ-Slide 15 Well 3D supports controlled matrix embedding with minimal reagent consumption.
When Do Organoids Start to Think?
One of the most fascinating questions is how far this complexity can go. At what point does a cluster of cells stop being just a model and start behaving like a network?
Brain organoids do not think in a conscious sense. But they do something remarkable. They generate spontaneous electrical activity, form synchronized firing patterns, and begin to organize into functional networks. In a way, they are not thinking, but they are starting to “talk” to each other.
These early interactions offer a unique glimpse into how the brain builds itself. Researchers can observe how signals emerge, how connections stabilize, and how network behavior evolves over time. It transforms organoids into living test systems where the first steps toward function can be studied in real time.
At the same time, organoids come with challenges. The lack of vascularization limits nutrient supply and growth, and variability between samples can affect reproducibility. Careful handling and stable culture conditions are therefore essential for generating consistent and reliable results.
From Structure to Function: Why the Microenvironment Matters
Building structure is only part of the story. To truly understand neuronal behavior, the surrounding microenvironment must also be controlled. Cells respond to physical and chemical cues such as flow, gradients, and spatial organization, which shape how networks form and function.
In the nervous system, endothelial cells are exposed to constant fluid flow, generating shear stress that influences cell morphology, signaling, and barrier integrity. In vitro, controlled flow enables the study of processes such as blood-brain barrier function and dysfunction.
Microfluidic systems enable precise control over flow, gradients, and nutrient exchange, adding dynamic conditions to otherwise static cultures and improving physiological relevance [7]. Platforms such as theµ-Slide I Luer 3D and µ-Slide Spheroid Perfusion integrate 3D culture with controlled flow for reproducible experiments.
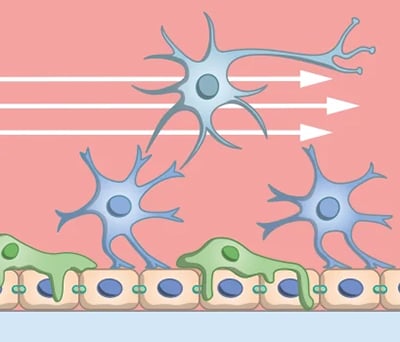
Fig. 3 | Controlled flow in microfluidic systems introduces shear stress and gradients, enabling more physiologically relevant cell behavior and blood-brain barrier function in vitro.
Micropatterning technologies bring an additional level of control to neuronal models by defining where neurons attach and how they connect. By guiding neurite outgrowth along defined geometries such as lines or confined islands, researchers can standardize network formation and create reproducible neuronal circuits to study connectivity, signal propagation, and cell-cell interactions.
Together, these approaches transform in vitro models from static structures into dynamic systems in which function emerges from the interaction between cells and their environment.
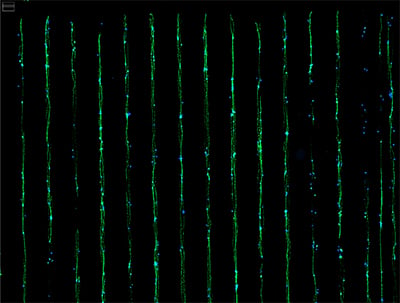
Fig. 4 | Human RealDRG grown on the µ-Slide VI 0.4 µ-Pattern ibiTreat lin20, pit170. The slide was pre-coated with silk for 2 hours at 37 °C before seeding the cells. After incubating the cells for 72 hours, fixation and staining with MitoTracker and DAPI were applied. Image courtesy of the group of Moran Amit at MD Anderson Cancer Center, University of Texas.
Conclusion: One System Is Not Enough
There is no single, perfect in vitro model of the nervous system. Each system captures a different level of complexity, from isolated cellular mechanisms to tissue-like organization and controlled interactions.
The real power lies in combining these approaches. Starting with 2D systems for mechanistic insights, moving into 3D cultures for physiological relevance, and integrating dynamic environments allows researchers to build a more complete understanding of neuronal biology.
As these technologies continue to evolve, they are not only improving how we study the brain but also redefining what is possible in neuroscience research.
References
- Wu, Y.-Y., et al., Opportunities and challenges for the use of induced pluripotent stem cells in modelling neurodegenerative disease. Open Biology, 2019. 9(1).
- Zhu, H., et al., Recent advances in 3D models of the nervous system for neural regeneration research and drug development. Acta Biomaterialia, 2025. 202: p. 1-26.
- Menduti, G. and M. Boido, Recent Advances in High-Content Imaging and Analysis in iPSC-Based Modelling of Neurodegenerative Diseases. Int J Mol Sci, 2023. 24(19).
- Zehtabian, A., et al., Automated Analysis of Neuronal Morphology in 2D Fluorescence Micrographs through an Unsupervised Semantic Segmentation of Neurons. Neuroscience, 2024. 551: p. 333-344.
- Li, Y., et al., Advances and Applications of Brain Organoids. Neurosci Bull, 2023. 39(11): p. 1703-1716.
- Bose, R., S. Banerjee, and G.L. Dunbar, Modeling Neurological Disorders in 3D Organoids Using Human-Derived Pluripotent Stem Cells. Front Cell Dev Biol, 2021. 9: p. 640212.
- Oddo, A., et al., Advances in Microfluidic Blood-Brain Barrier Models. Trends Biotechnol, 2019. 37(12): p. 1295-1314.
 (0)
(0)  (0)
(0)




