Micropatterning 101
ibidi Blog | February 26, 2026 | Abhishek Derle, ibidi GmbH
Cells rarely behave identically even when cultured under identical conditions. In a standard dish, some cells spread broadly while others remain compact. Some polarize and migrate, whereas others divide in place. Variability in cell shape, adhesion area, and local cell-to-cell contacts directly contributes to differences in signaling, migration speed, differentiation potential, drug response, and even cell survival and fate [1, 2]. We often attribute this to biological heterogeneity, and part of it certainly is, but part of it is simply spatial randomness.
Micropatterning addresses this at its root.
In this blog, we will provide a practical overview of micropatterning, including how the technology evolved, how different geometries are used in research, and why spatial control is becoming increasingly important in modern cell biology.
What Micropatterning Actually Means?
Micropatterning is a technique that precisely defines where cells can attach. Rather than providing a single large adhesive surface, it creates small adhesive regions on a passivated background. The regions outside the patterned areas are engineered to inhibit both cell attachment and protein binding, effectively confining cellular attachment to the intended geometric shapes.
These adhesive regions can be circular, square, linear, or custom-designed, and they are defined in micrometer dimensions. By adjusting pattern size and spacing, researchers can control how many cells attach per spot, how far apart neighboring cells are positioned, and whether cells remain isolated or assemble into clusters. Geometry becomes an experimental parameter rather than an uncontrolled variable, an idea that has been central to the field of cell morphogenesis and cytoskeletal organization [3].
The concept has evolved considerably over time. Early approaches relied on stamping extracellular matrix proteins onto culture surfaces using elastomer molds. These methods were powerful but required technical expertise and careful optimization. Advances in surface chemistry improved background passivation and pattern stability, making confinement more reliable. Today, ready-to-use micropatterned substrates eliminate fabrication steps and provide sterile, imaging-optimized platforms that make spatial control accessible to any cell biology laboratory, with recent developments expanding flexibility and engineering precision [4].
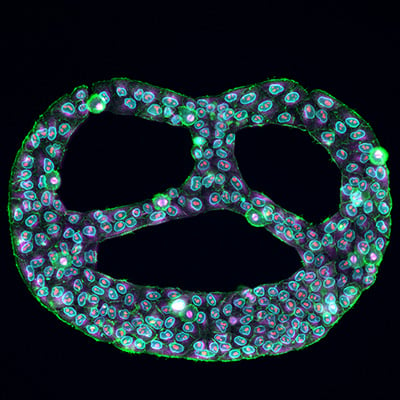
Fig. 1 | A Pretzel Under a Microscope. Multiplex immunostaining of HeLa cells grown on Custom µ-Slide VI 0.4 µ-Pattern ibiTreat
Geometry as a Practical Tool
The real strength of micropatterning lies in its ability to standardize spatial organization without complicating experimental workflows. By choosing specific geometries, researchers can tailor the setup to address distinct biological questions.
Single Cell Arrays
Small adhesive regions with defined spacing allow individual cells to attach separately from one another. This setup is especially useful in imaging-based assays, where overlapping cells complicate segmentation and quantification. Organized single cell arrays improve image clarity and make automated analysis more reliable. For studies that depend on precise morphological measurements or fluorescence intensity analysis, spatial separation alone can significantly improve data quality.
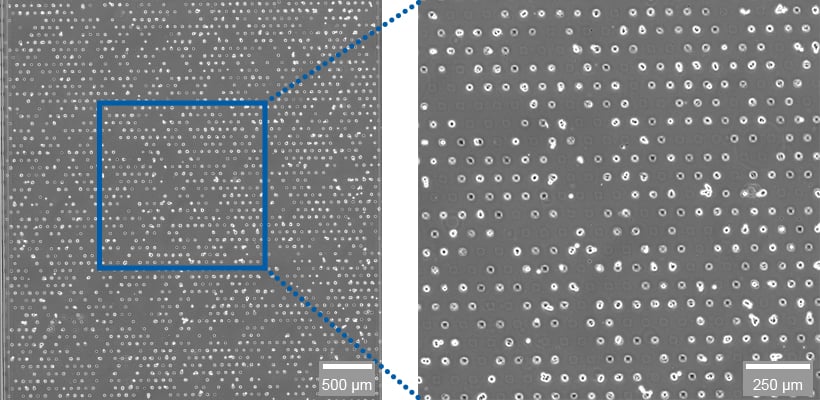
Fig. 2 | A549 cells grown on a laminin-coated µ-Slide VI 0.4 µ-Pattern ibiTreat, sqr30, pit75, hex.
Multi Cell Arrays
Larger adhesive areas allow several cells to attach within one defined region. Because the surrounding surface prevents unwanted adhesion, cells accumulate only within the patterned spot, forming reproducible micro colonies. This controlled clustering is valuable in studies of cell-to-cell communication, tumor microenvironment models, and immune cell interaction assays, where a consistent spatial arrangement simplifies comparison across experiments. Defined colony geometries have even been used to reproduce early embryonic spatial organization in vitro [5], highlighting how geometry alone can influence collective behavior.
Video 1 | RCC-26 cancer cells immobilized on multi-cell pads (µ-Slide VI 0.4 µ-Pattern ibiTreat, cir200, pit600, hex). Effector T cells applied in collagen I matrix (Collagen Type I, Rat Tail) induce apoptotic body formation of cancer cells.
Spheroid Formation
Micropatterning can also support controlled spheroid generation. When cells are seeded in suspension onto defined adhesive spots surrounded by a fully non-adhesive background, they form compact, uniform aggregates. This reduces variability between spheroids and improves reproducibility in screening and drug testing applications. Instead of dealing with heterogeneous aggregate sizes, researchers can work with standardized three-dimensional structures, similar in concept to matrix-controlled organoid systems [6].
Line Arrays
Linear patterns create micro-lane environments that guide cell orientation and directional growth. Cells align along the geometry of the line, which is useful for studying neurite extension, cytoskeletal organization, or structured migration. One-dimensional confinement has been shown to better mimic fibrillar in vivo migration compared to unconstrained 2D culture [7]. By reducing random orientation, line arrays make it easier to analyze directional processes and intracellular transport with greater consistency.
Across all these formats, micropatterning does not change the biology itself. It simply removes uncontrolled spatial differences that can obscure interpretation.
Video 2 | HT-1080 cells grown on a µ-Slide 8 Well high µ-Pattern ibiTreat, lin20, pit170 coated with Laminin.
Micropatterning Under Flow
Cell culture in vivo is rarely static. Tissues experience continuous fluid movement, nutrient gradients, and mechanical forces. Micropatterning can be combined with perfusion systems to introduce these dynamic conditions into controlled in vitro experiments.
When patterned cells or aggregates are cultured within perfusable slide systems, continuous media circulation provides stable nutrient supply and defined shear stress. Structured spheroids maintained under flow can exhibit improved compactness and long-term stability, while adherent cells experience mechanical stimulation without losing their spatial organization. By combining geometric confinement with fluid dynamics, researchers can explore how spatial arrangement and mechanical cues interact in a more physiologically relevant context. This integration moves micropatterning beyond static confinement and closer to tissue-like experimental environments.
Create Your Own Pattern
Not every research question fits into predefined geometries. Different experimental designs may require specific spatial arrangements, separated interaction zones, or unique dimensions that are not available as standard formats.
Custom micropatterning allows researchers to design geometry around their scientific question. Whether the goal is to create compartmentalized co-culture systems, structured cellular networks, or defined interaction interfaces, customization ensures that spatial organization supports the biological objective.

Fig. 3 | You need support or want to test the Custom ibiTreat µ-Patterns? Contact us at: info@ibidi.com or choose up to three free samples from our ibiTreat micropatterning standard products!
In this sense, micropatterning becomes more than a technique for controlling adhesion. It becomes part of experimental design. By tailoring geometry to the needs of the assay, researchers can integrate spatial control directly into their study logic rather than adapting their experiment to a fixed format.
Why It Matters Today and What Comes Next
Modern cell biology relies heavily on imaging, quantification, and reproducibility. High content analysis, artificial intelligence-based image processing, and spatial omics technologies all benefit from standardized cell organization. Micropatterning reduces spatial noise and improves comparability across wells, time points, and laboratories.
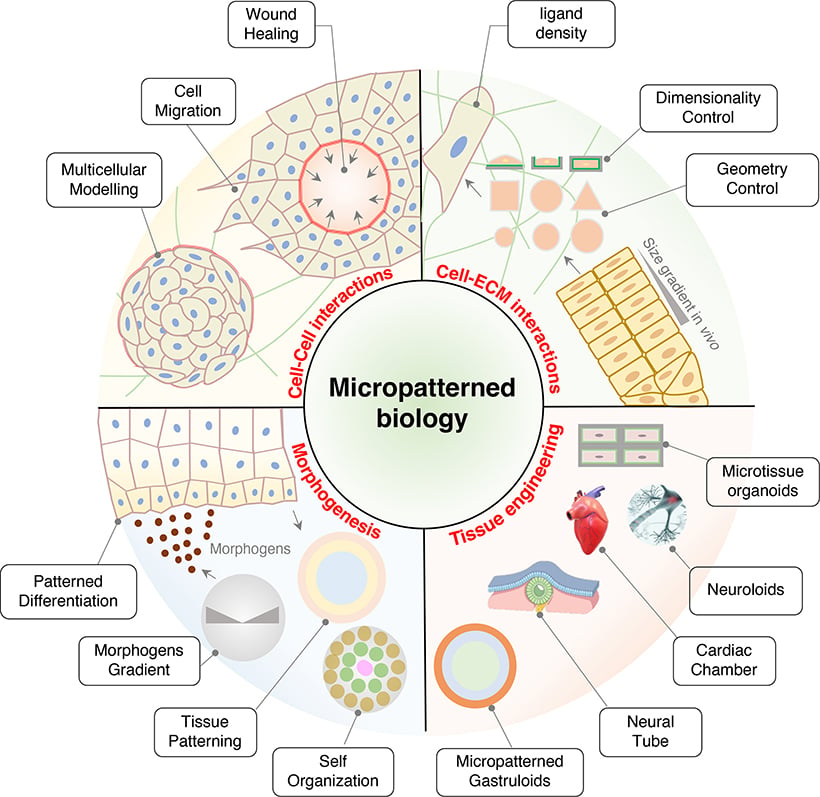
Fig. 4 | The application of micropatterned biology. Adapted from Hu and Bao, 2024 [4].
In immunology, defined target cell arrays simplify live cell interaction assays. In toxicology and drug development, uniform spheroid formation supports reproducible screening workflows. In neuroscience, guided microlanes clarify directional growth patterns. Across disciplines, spatial control enhances clarity.
As experimental systems become more complex, controlling geometry at the micrometer scale will likely become even more important. What started as a way to simply constrain where cells attach has become a creative approach for designing the very architecture of experiments. Micropatterning now empowers scientists to shape the landscape of their studies with precision and imagination.
Sometimes, clearer biology begins with something as small as a few micrometers of control.
References
- Chen, C.S., et al., Geometric control of cell life and death. Science, 1997. 276(5317): p. 1425-1428.
- McBeath, R., et al., Cell shape, cytoskeletal tension, and RhoA regulate stem cell lineage commitment. Dev Cell, 2004. 6(4): p. 483-495.
- Théry, M., Micropatterning as a tool to decipher cell morphogenesis and functions. J Cell Sci, 2010. 123(Pt 24): p. 4201-4213.
- Hu, X. and M. Bao, Advances in micropatterning technology for mechanotransduction research. Mechanobiology in Medicine, 2024. 2(3): p. 100066.
- Warmflash, A., et al., A method to recapitulate early embryonic spatial patterning in human embryonic stem cells. Nat Methods, 2014. 11(8): p. 847-854.
- Gjorevski, N., et al., Designer matrices for intestinal stem cell and organoid culture. Nature, 2016. 539(7630): p. 560-564.
- Doyle, A.D., et al., One-dimensional topography underlies three-dimensional fibrillar cell migration. J Cell Biol, 2009. 184(4): p. 481-490.
 (1)
(1)  (0)
(0)




