Light Sheet Fluorescence Microscopy (LSFM)
Applications:
LSFM is applied for live cell 3D imaging of thick biological samples as a whole, such as embryos, spheroids, organoids, and whole animals. The strongly reduced background signal, minimized phototoxicity, and increased scanning speed make LSFM superior to epifluorescent microscopy for the analysis of thick living specimens.
Principle:
Researchers can choose between several LSFM/SPIM (Single/Selective Plane Illumination Microscopy) systems that differ in certain aspects but all have the following main features: in contrast to confocal or widefield fluorescent microscopy only a thin section of the specimen is illuminated by a light sheet. Another important difference is the separation of the optical pathways of illumination and detection. To avoid the measurement of out-of-focus fluorescence, emitted light is detected on a different axis to the illumination (e.g., orthogonal). By doing this, unnecessary out-of-focus fluorescence is not excited, which prevents photobleaching and photodamage in regions that are currently not being scanned. Finally, with LSFM, the image is created by scanning a plane of light (optical sectioning), instead of point by point, which markedly increases scanning speed when compared to confocal microscopy.
These advantages—reduced cellular stress, background fluorescence, and time consumption—are especially important when doing 3D live cell imaging of sensitive biological samples.
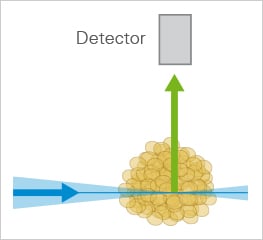
The principle setup of LSFM.
ibidi Solutions:
Please note: The suitability of the ibidi labware for LSFM depends on the well/channel geometry, the used LSFM system and its optical setup. It has to be checked individually before each experiment. |  |




