Confocal Microscopy:
Principle, Advantages, and Applications
Confocal microscopy is a fluorescence imaging technique used to visualize cells and tissues with high contrast. It is especially useful for imaging thick samples such as spheroids, organoids, and tissue sections. By eliminating out-of-focus light through optical sectioning, confocal microscopy enables sharp, high-contrast images of specific focal planes. In cell biology and biomedical research, it is widely applied for studying cell morphology, protein localization, and dynamic processes in both fixed and living cells.
What Is Confocal Microscopy?
Confocal laser scanning microscopy (CLSM) is an advanced fluorescence microscopy method that uses a focused laser beam and a pinhole aperture to collect light from a defined focal plane. The pinhole blocks most out-of-focus fluorescence, improving image contrast and resolution. This selective detection allows optical sectioning, meaning that thin slices of a sample can be imaged individually. By stacking these images (z-stacks), researchers can reconstruct detailed 3D models of biological structures such as cells, spheroids, or tissues.
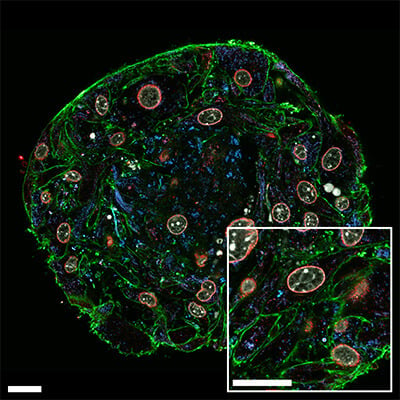
Confocal image of a NIH3T3 spheroid grown on the µ-Slide VI 0.4 µ-Pattern ibiTreat, cir200, pit600, hex, labeled with abberior dyes and imaged with the abberior STEDYCON. Scale bar: 20 µm.
When to Use Confocal Microscopy
Confocal microscopy is the preferred method when:
- Imaging thick samples (e.g., spheroids, organoids, tissues)
- High-resolution 3D reconstruction is required
- Studying protein localization using fluorescence labeling
- Performing multicolor imaging of multiple targets
- Reducing background signal from out-of-focus light
For larger samples or in vivo imaging, alternative techniques such as light sheet microscopy and two-photon microscopy are needed.
How Does Confocal Microscopy Work?
Confocal microscopy uses a focused laser beam that scans across the sample point by point. Unlike widefield fluorescence microscopy, which illuminates the entire sample, using CLSM only fluorophores within a small focal volume are excited. The emitted fluorescence passes through a pinhole aperture that blocks out-of-focus light. This results in high-contrast images with improved resolution compared to widefield microscopy.
By acquiring a series of images along the z-axis, also called z-stack imaging, researchers can reconstruct three-dimensional (3D) structures of cells, spheroids/organoids, or tissue slices.
Modern systems also enable multicolor imaging by using multiple lasers and detectors. Spectral unmixing techniques separate overlapping fluorescence signals, allowing accurate visualization of multiple cellular structures simultaneously.
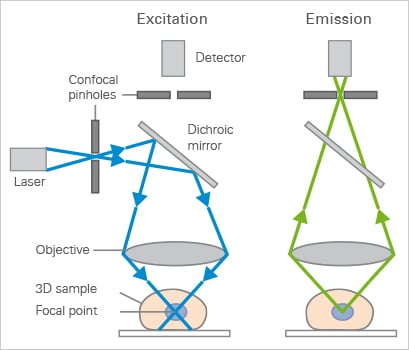
Excitation (left) and emission (right) light path in a basic confocal microscope setup.
Comparison: Widefield vs Confocal Microscopy
Widefield and confocal microscopy are the most commonly compared fluorescence imaging techniques, each suited for different sample types and imaging goals.
When to use widefield:
- Fast imaging of dynamic processes (e.g., cell migration, mitosis)
- Thin samples such as cell monolayers
- Live-cell imaging with low phototoxicity
- Routine fluorescence and multicolor imaging
When to use confocal:
- Thick samples or tissues
- 3D imaging and z-stack acquisition
- High-contrast imaging with reduced background
- Precise optical sectioning
| Feature | Widefield | Confocal |
| Illumination | Full field | Point scanning |
| Detection | Camera-based | Detector with pinhole |
| Speed | Very fast | Slower |
| Sensitivity | High | Moderate |
| Optical sectioning | No | Yes |
| Out-of-focus light | Present | Strongly reduced |
| Sample thickness | Thin samples | 3D samples |
| Live cell imaging | Excellent | Possible |
Advantages and Limitations of Confocal Microscopy
Confocal microscopy provides high-resolution imaging with reduced background fluorescence by rejecting most out-of-focus light. It enables optical sectioning of thick samples and supports 3D reconstruction through z-stacks, as well as multicolor imaging of multiple targets.
However, confocal microscopy has limitations in light penetration depth (typically up to 100 µm). Moreover, it can cause strong photobleaching or even phototoxicity effects, a factor that is especially important when imaging living samples
What Are the Main Applications Using Confocal Microscopy?
Confocal microscopy is widely used in biological and biomedical research for high-resolution 3D imaging. In 3D cell culture models such as spheroids and organoids, it enables analysis of cellular organization, viability, and structural complexity. In cancer research, it is applied to study tumor architecture, viability gradients, and drug responses in tissue-like models.
In neuroscience, confocal microscopy allows precise visualization of neuronal networks and synaptic structures. It is also used in cell migration assays to track dynamic behavior and in protein localization studies to analyze intracellular distribution. Additional applications include live cell time-lapse imaging and detailed tissue imaging, where optical sectioning is essential for analyzing thick samples.
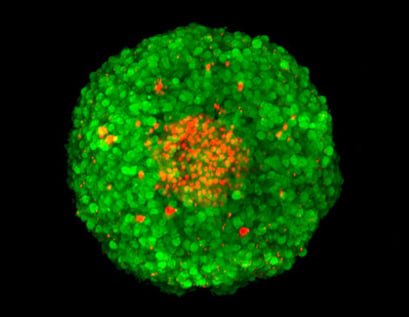
Z-stack image of an FDA/PI-stained MCF-7 spheroid, acquired using confocal microscopy. Green: FDA-stained living cells. Red: PI-stained dead cells in the necrotic center of the spheroid.

ibidi Solutions for Confocal Imaging
All ibidi µ-Slides, µ-Dishes, and µ-Plates are fully compatible with confocal and high-resolution fluorescence microscopy. The ibidi #1.5 Polymer Coverslip and the ibidi #1.5H Glass Coverslip provide ideal optical conditions for confocal microscopy.
Common Problems and Troubleshooting
Why is my fluorescence signal so weak?
A weak signal is often caused by low fluorophore concentration, poor staining, or inefficient excitation.
Solution: Increase laser power or optimize staining first. If the signal is still too low, you can open the pinhole beyond ~1 Airy unit to collect more light. However, this reduces optical sectioning and increases background, meaning your image will lose contrast and axial resolution.
Why is the background signal so high?
Background fluorescence can result from out-of-focus light or non-specific staining.
Solution: Adjust the pinhole to e.g. ~1 Airy unit for optimal optical sectioning, and improve your staining protocol, use well-validated antibodies or dyes, aliquot reagents to avoid contamination, use fresh reagents and mounting medium, use high-quality imaging substrates such as optical-grade coverslips or ibidi polymer coverslips.
Why do I see photobleaching during imaging?
Prolonged or intense excitation light damages fluorophores, especially when not mounted with anti-fade media.
Solution: Use lower laser power and compensate with signal averaging instead of increasing intensity. Additionally, choose more photostable fluorophores and minimize exposure time to preserve fluorescence signal.
Why is my image drifting in z-direction?
Z-drift is typically caused by temperature fluctuations or mechanical instability during imaging.
Solution: Use stable mounting, temperature control, and environmental chambers for live cell imaging.
Why do my fluorescence channels overlap?
Spectral overlap occurs when the emission spectra of different fluorophores overlap, causing signal from one fluorophore to be detected in multiple channels.
Solution: Choose fluorophores with well-separated emission spectra, adjust detection windows, use sequential scanning, or apply spectral unmixing to accurately separate the signals.
FAQs
What is the principle of confocal microscopy?
Confocal microscopy uses a focused laser and a pinhole to block out-of-focus light, enabling high-resolution optical sectioning of samples.
What is confocal microscopy used for?
It is used for high-resolution imaging of cells, tissues, and 3D structures such as spheroids and organoids.
Can confocal microscopy be used for live cells?
Yes, but phototoxicity must be minimized by optimizing laser power, exposure time, and environmental conditions.
What is spinning disk confocal microscopy?
Spinning disk confocal microscopy uses multiple pinholes on a rotating disk to scan the sample simultaneously, enabling much faster imaging compared to point-scanning confocal systems. It is ideal for live-cell imaging and dynamic processes with reduced phototoxicity, but provides slightly lower optical sectioning and resolution compared to conventional confocal microscopy.
What is Airyscan detection in confocal microscopy?
Airyscan detection is an advanced confocal imaging approach that uses a detector array to capture more spatial information from emitted light, improving resolution and signal-to-noise ratio. It enables higher sensitivity and better image quality compared to standard confocal microscopy, especially for low-signal or high-resolution applications.
Which labware is best for confocal microscopy?
All ibidi labware is fully compatible with confocal and high-resolution fluorescence microscopy. Find more information here.





