Fluorescence Recovery After Photobleaching (FRAP)
Fluorescence Recovery After Photobleaching (FRAP) is a fluorescence microscopy technique used to study molecular mobility and diffusion in live cells. It involves photobleaching a region of interest and measuring fluorescence recovery to analyze protein dynamics.
ibidi supports FRAP experiments with high-optical-quality labware optimized for live cell imaging and accurate fluorescence measurements.
Principle and Applications of FRAP: Simply Explained
FRAP Principle: Understanding Fluorescence Recovery After Photobleaching
Fluorescence Recovery After Photobleaching (FRAP) is commonly performed using a modern confocal microscope and requires fluorescent labeling of the molecule of interest, such as with a GFP-tagged antibody. A typical FRAP experiment involves three key steps: first, the baseline fluorescence intensity is recorded in a defined region of interest; next, targeted photobleaching is performed by focusing a laser to extinguish fluorescence in that area. As a result, a dark area in the otherwise fluorescent sample can be observed. Finally, fluorescent molecules from the surroundings can diffuse through the sample and exchange the photobleached molecules with intact ones. This fluorescence recovery is measured over time.
This recovery process provides valuable data about molecular diffusion and protein mobility. Accurate analysis of FRAP data requires careful planning, as recovery kinetics can be influenced by numerous experimental factors. Mathematical modeling of the fluorescence recovery curve enables calculation of the diffusion coefficient and other mobility parameters.
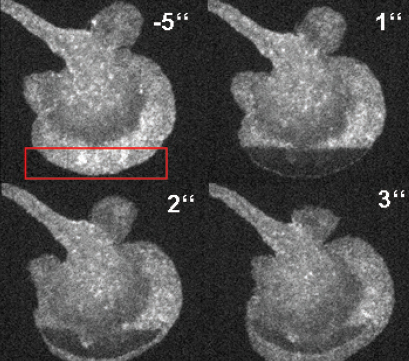
FRAP-based visualization of the F-actin diffusion in a primary dendritic cell.
FRAP Applications in Live Cell Imaging and Molecular Mobility Studies
FRAP is a powerful fluorescence microscopy technique used in live cell imaging to study molecular mobility and protein dynamics in real time. It is widely applied to analyze membrane protein diffusion, cytoskeletal interactions, and intracellular transport mechanisms.
In addition to fundamental research, FRAP plays a key role in biomolecular interaction analysis and drug discovery assays, where understanding protein trafficking and fluorescent protein-tagged constructs is essential for evaluating therapeutic targets and cellular responses.
H.C. Ishikawa-Ankerhold, R. Ankerhold, G.P.C. Drummen. Advanced Fluorescence Microscopy Techniques—FRAP, FLIP, FLAP, FRET and FLIM. Molecules, 2012, 10.3390/molecules17044047
Read article
ibidi SolutionsThe ibidi Polymer Coverslip and the ibidi Glass Coverslip provide ideal optical conditions for fluorescence microscopy. |  |
FRAP is possible without restrictions when using any of the ibidi labware that contains the ibidi Polymer Coverslip or the ibidi Glass Coverslip bottom.




