Widefield Fluorescence Microscopy:
Principle, Applications, and When to Use It
Widefield fluorescence microscopy is a widely used imaging technique for visualizing labeled structures in cells and tissues. It enables fast image acquisition, high sensitivity, and straightforward multicolor imaging, making it especially useful for live cell imaging, immunofluorescence, and time-lapse experiments. Because the entire field of view is illuminated at once, widefield systems are ideal for thin samples and dynamic processes that need rapid capture. In cell biology, the method is commonly used to study protein localization, cytoskeletal organization, cell migration, mitosis, and fluorescent reporter expression. For thicker specimens, however, out-of-focus light can reduce contrast, which is why confocal microscopy is often the better choice.
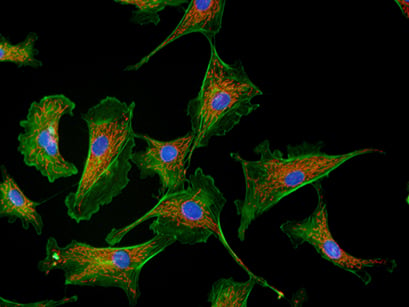
Widefield immunofluorescence image of bovine aortic endothelial cells (BAOECs) showing F-actin cytoskeleton (green, Alexa488–phalloidin), mitochondria (red, MitoTracker Red), and nuclei (blue, DAPI), acquired with a 63× oil immersion objective.
What Is Widefield Fluorescence Microscopy?
Widefield fluorescence microscopy, often referred to as epifluorescence microscopy, is one of the most widely used imaging techniques in modern cell biology. It enables the visualization of fluorescently labeled structures in cells and tissues. In this method, the entire field of view is illuminated at once, allowing fluorophores in the sample to be excited simultaneously. The emitted fluorescence is then detected using a camera, making the technique fast and sensitive and therefore well suited for live cell imaging, time-lapse experiments, and high-content screening. It is particularly suitable for thin samples such as cell monolayers and is widely used in both fixed and live-cell imaging experiments.
When to Use Widefield Fluorescence Microscopy
Use widefield fluorescence microscopy when:
- you need fast image acquisition for dynamic processes
- you work with thin samples (e.g., adherent cells)
- you perform multicolor fluorescence imaging
- you want a sensitive, camera-based detection method
- you conduct routine immunofluorescence experiments
- optical sectioning is not required
Choose confocal microscopy instead when:
- imaging thick or 3D samples
- out-of-focus blur reduces image quality
- optical sectioning is required

Want to image specimens without disturbing out-of-focus light?
Learn more about confocal microscopy and when to use it.
Which Samples Can Be Imaged With Widefield Fluorescence Microscopy?
Widefield fluorescence microscopy works best for thin samples: Because the entire sample volume is illuminated, fluorophores both inside and outside the focal plane are excited simultaneously. As a result, only thin specimens, such as monolayers of cultured cells or thin tissue sections, can be imaged with high clarity. Thicker samples typically require physical sectioning
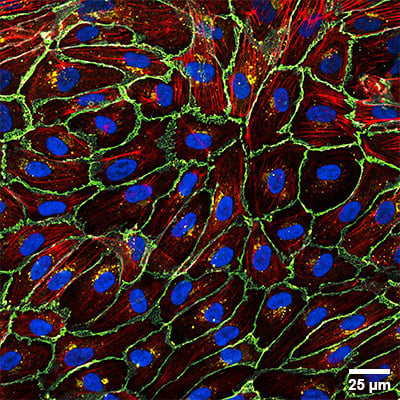
Widefield fluorescence microscopy images of a single layer of adherent cells. Human umbilical vein endothelial cells (HUVEC) cultured in the µ-Slide I Luer and stained after fixation. Colors indicate VE-cadherin (cyan), Connexin 43 (yellow), F-actin (red), and nuclei (blue). Scale bar: 25 µm.
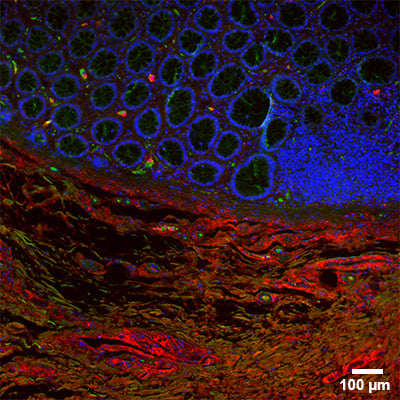
Widefield fluorescence microscopy image of a tissue section. Mouse intestinal formalin-fixed paraffin-embedded (FFPE) tissue section stained in the sticky-Slide Tissue. Cell nuclei are labeled with DAPI (blue), F-actin with Phalloidin-iFluor 488 (green), and collagen type I with Alexa Fluor 647 (red). Scale bar: 100 µm.
Comparison: Widefield vs Confocal Microscopy
Widefield and confocal microscopy are the most commonly compared fluorescence imaging techniques, each suited for different sample types and imaging goals.
When to use widefield:
- Fast imaging of dynamic processes (e.g., cell migration, mitosis)
- Thin samples such as cell monolayers
- Live-cell imaging with low phototoxicity
- Routine fluorescence and multicolor imaging
When to use confocal:
- Thick samples or tissues
- 3D imaging and z-stack acquisition
- High-contrast imaging with reduced background
- Precise optical sectioning
| Feature | Widefield | Confocal |
| Illumination | Full field | Point scanning |
| Detection | Camera-based | Detector with pinhole |
| Speed | Very fast | Slower |
| Sensitivity | High | Moderate |
| Optical sectioning | No | Yes |
| Out-of-focus light | Present | Strongly reduced |
| Sample thickness | Thin samples | 3D samples |
| Live cell imaging | Excellent | Possible |
How Does Widefield Fluorescence Microscopy Work?
In widefield fluorescence microscopy, excitation light passes through the objective lens and illuminates the entire sample. Fluorophores absorb this light and emit fluorescence at longer wavelengths. The emitted light is separated from the excitation light using a dichroic mirror and emission filters before being detected. Because the entire field is illuminated simultaneously, imaging is fast. However, fluorescence from out-of-focus planes is also collected, which can reduce contrast in thicker samples.
What Are the Key Components of a Widefield Fluorescence Microscope?
A typical widefield fluorescence microscope consists of several essential optical elements:
Excitation Light Sources
Widefield microscopes use either a broadband white light source (e.g., mercury or xenon arc lamps) combined with bandpass filters, or LED light engines providing spectrally separated excitation bands.
Optical Filters
- Excitation filter: Transmits only the desired wavelengths needed for fluorophore excitation.
- Dichroic mirror: An optical component with wavelength-dependent reflective and transmissive properties. In a microscope setup, it reflects the excitation light toward the specimen while transmitting the emitted fluorescence.
- Emission filter: Isolates fluorescence emission and blocks unwanted wavelengths.
Detectors
Highly sensitive cameras, such as sCMOS or CCD cameras, are typically used. These detectors enable high-speed imaging and quantitative fluorescence measurements.
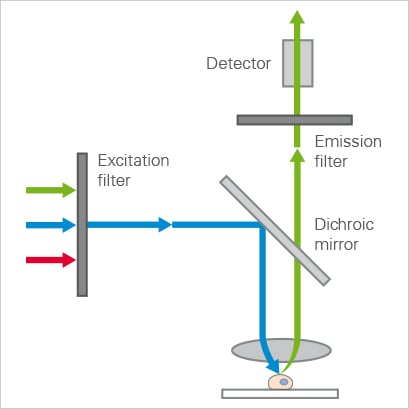
Schematic of a widefield setup showing an excitation and emission light path. The excitation filter transmits a narrow wavelength band, which is reflected by the dichroic mirror and focused on the sample. The red-shifted emission passes through the dichroic mirror and emission filter before reaching the detector.
What Are the Advantages and Limitations of Widefield Fluorescence Microscopy?
Widefield fluorescence microscopy offers several important advantages, such as a fast image acquisition, a high sensitivity due to camera detection, relatively low phototoxicity and compatibility with live cell imaging.
The main limitation of widefield fluorescence microscopy is the detection of out-of-focus fluorescence, which reduces image contrast, particularly in thicker specimens. Widefield systems provide limited optical sectioning capabilities, exhibit lower axial resolution than confocal microscopy, and therefore produce blurred images in tissues and three-dimensional samples.
What Are the Main Applications of Widefield Fluorescence Microscopy in Cell Biology?
Widefield fluorescence microscopy is a fundamental tool in biological research, enabling the visualization of cellular structures and molecular processes with high specificity and comparatively low phototoxicity.
Common applications include immunofluorescence imaging, where antibody-based labeling is widely used to detect proteins within cells. Another key application is the imaging of fluorescent proteins in expression cell lines, where genetically encoded fluorophores such as GFP, mCherry, or YFP allow visualization of protein localization and dynamics in living cells.
Widefield microscopy is particularly well suited for live cell imaging, enabling the observation of dynamic biological processes such as cell migration, vesicle trafficking, cytoskeletal remodeling, and mitosis. Because the entire field of view is recorded simultaneously, this technique supports rapid time-lapse imaging or high-content imaging.
What Is the Resolution of Widefield Fluorescence Imaging?
The lateral resolution of widefield fluorescence microscopy is limited by diffraction and typically ranges from approximately 200–300 nm, depending on the numerical aperture of the objective and the emission wavelength.
Because fluorescence originating from multiple focal planes contributes to the recorded image, thick specimens often appear blurred due to the accumulation of out-of-focus light. In contrast, thin samples (such as adherent cultured cells) generally allow imaging with high clarity. However, even adherent cells typically exceed 1 µm in thickness, which approaches the axial resolution limit of widefield systems and can therefore lead to residual out-of-focus background.
Computational methods such as image deconvolution can improve image quality by estimating and redistributing out-of-focus light based on the system’s point spread function (PSF), thereby enhancing contrast and apparent resolution.
Want to achieve higher resolution?
Learn how super-resolution microscopy techniques bypass the physical diffraction limit.

ibidi Solutions for Widefield Fluorescence Microscopy
All ibidi µ-Slides, µ-Dishes, and µ-Plates are fully compatible with widefield fluorescence microscopy. The ibidi #1.5 Polymer Coverslip and the ibidi #1.5H Glass Coverslip provide ideal conditions for cell cultivation and microscopy.
Common Problems and Troubleshooting
Why are my fluorescence images blurry?
Out-of-focus fluorescence from planes above and below the focal plane reduces image contrast, especially in thicker samples.
Solution: Use thinner samples (e.g., cell monolayers), use high NA objectives, apply deconvolution to improve contrast. If this is not enough, consider switching to confocal microscopy for thick specimens.
Why is the background signal so high?
Non-specific staining, autofluorescence, or poor optical quality of imaging materials can lead to a higher background signal.
Solution: Improve staining protocols and washing steps, use well-validated antibodies or dyes, aliquot reagents to avoid contamination, use fresh reagents and mounting medium, use high-quality imaging substrates such as optical-grade coverslips or ibidi polymer coverslips.
Why is my fluorescence signal so weak?
A weak signal can either originate from a low fluorophore brightness, insufficient staining, photobleaching, or suboptimal imaging settings.
Solution: Use brighter fluorophores, higher labeling density or longer incubation times, optimize exposure time and illumination intensity, increase numerical aperture (NA) of the objective.
Why do I see photobleaching during imaging?
Prolonged or intense excitation light damages fluorophores, especially when not mounted with anti-fade media.
Solution: Reduce light intensity and exposure time, use more photostable fluorophores, minimize acquisition frequency in time-lapse experiments, use appropriate imaging buffers or antifade reagents.
FAQs
What Is the Difference Between Widefield and Epifluorescence Microscopy?
In practice, the terms are often used interchangeably. Epifluorescence describes the optical geometry in which excitation and emission light travel through the same objective lens (epi-illumination). Widefield microscopy refers to the imaging mode in which the entire field of view is illuminated and detected simultaneously using a camera.
Which Objectives Are Best for Widefield Fluorescence?
Objectives with a high numerical aperture (NA) are preferred because they improve resolution, brightness, and photon collection efficiency. Oil immersion objectives (NA ≥ 1.3) provide optimal performance for high-resolution imaging, while water immersion objectives are advantageous for live cell imaging due to refractive index matching and reduced spherical aberration.
Plan Apochromat objectives are recommended for fluorescence applications because they provide superior chromatic correction across multiple wavelengths, flat field correction for camera-based imaging and high transmission efficiency.
The optimal magnification depends on the camera pixel size and the required sampling (Nyquist criterion).
Which Fluorophores Can Be Used for Widefield Imaging?
Widefield fluorescence microscopy is compatible with a broad range of fluorophores. Fluorophore selection depends on excitation source compatibility, filter sets, photostability, brightness, and spectral separation for multicolor imaging.
How Can Out-of-Focus Blur Be Reduced?
Out-of-focus blur in widefield fluorescence microscopy arises from fluorescence emitted outside the focal plane. Several strategies can reduce this effect:
Optical approaches
- Imaging thinner specimens
- Using objectives with higher NA
- Refractive index matching of immersion media
- Reducing background fluorescence through proper sample preparation
Computational approaches
- Image deconvolution using the system’s point spread function (PSF)
- 3D deconvolution for z-stacks to improve optical sectioning
Alternative imaging modalities
- Confocal or non-linear microscopy techniques for physical optical sectioning
Is Widefield Microscopy Suited for 3D Samples?
Not ideally. Widefield fluorescence microscopy collects fluorescence from all focal planes, which leads to out-of-focus blur and reduced contrast in thick or 3D specimens. For such samples, techniques like confocal, light-sheet, or multiphoton microscopy provide optical sectioning and improved 3D imaging.
What is the Difference Between Widefield vs. Confocal Microscopy?
In widefield fluorescence microscopy, the entire specimen is illuminated simultaneously, and fluorescence from all focal planes is collected by the detector. This allows fast image acquisition and high sensitivity, but out-of-focus light reduces image contrast and limits axial resolution, especially in thick or 3D samples.
In confocal microscopy, illumination is spatially confined to a single point (or a small spot) using a focused laser beam, often with coherent light. A pinhole in front of the detector blocks fluorescence originating from out-of-focus planes, enabling optical sectioning and improved contrast and axial resolution.
Is Widefield or Confocal Imaging More Phototoxic for the Sample?
Phototoxicity in fluorescence microscopy depends on multiple factors, including illumination intensity, exposure time, and imaging modality. While widefield microscopy often uses lower illumination intensities, it exposes the entire sample volume, potentially causing photobleaching outside the focal plane. In contrast, confocal microscopy uses a point-scanning approach and illuminates only a confined focal spot at a given time, thereby restricting light exposure largely to the focal plane. However, because the excitation light is concentrated within a small volume, the local laser intensity, and thus the potential phototoxic stress at the focal spot, is higher.
References
J. B. Pawley. Handbook of Biological Confocal Microscopy. Springer. 2006, doi: 10.1007/978-0-387-45524-2
J. R. Lakowicz. Principles of Fluorescence Spectroscopy. Springer. 2006, doi: 10.1007/978-0-387-46312-4





