Transfection Methods: Viral Transduction
The term "transduction" is used to describe a virus-mediated transfer of nucleic acids into cells. In contrast to transfection of cells with foreign DNA or RNA, no transfection reagent is needed here. The viral vector, itself, also called virion, is able to infect cells and transport the DNA directly into the nucleus, independent of further actions. After the release of the DNA into the nucleus, the protein of interest is produced using the cells' own machineries.
Method Comparison
| Adenoviral transduction | Lentiviral transduction | Transfection | |
| Viral genome material | dsDNA | RNA | - |
| Transgene expression | Transient | Stable | Transient or stable |
| Genome integration | Episomal | Integration into host genome | Dependent on transfection method |
| Cell types | Dividing and non-dividing cells | Dividing and non-dividing cells | Dividing and non-dividing cells, dependent on transfection method |
| Efficiency | Up to 100% transduction efficiency | Up to 100% transduction efficiency | Variable transfection efficiency |
| Biosafety level | 2 | 2 | 1 |
Adenoviral vectors have proven to be a very successful transduction tool in many eukaryotic cell types, such as human and rodent cells. Besides dividing cell lines, this method gives access to difficult-to-transfect cells, such as primary cells. Adenovirus-mediated transduction is always transient, meaning that no nucleotide integration into the host genome occurs. Transduction efficiencies of up to 100% can easily be achieved. Importantly, biosafety level S2 is needed for adenoviral transduction.
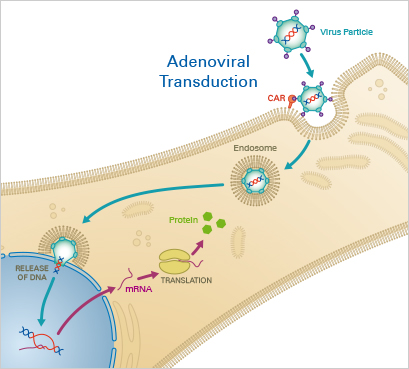
Principle:
Adenoviruses are a class of double stranded DNA viruses that efficiently deliver nucleotides directly into target cells. After initial binding to the Coxsackievirus and adenovirus receptor (CAR), which is expressed on the cell membrane, the virus enters the host cell via endocytosis. Following endosomal escape, the viral genome is transported into the nucleus, where it is expressed by the replication machineries of the host cell. In contrast to other viruses, the adenoviral DNA remains episomal, i.e., it is not integrated into the host genome. Nowadays, replication-deficient adenoviruses are widely used for transduction and gene therapy, due to their high efficiency and low pathogenicity.
Lee C.S., et al. Adenovirus-mediated gene delivery: Potential applications for gene and cell-based therapies in the new era of personalized medicine. Genes Dis, 2017, 10.1016/J.GENDIS.2017.04.001
read abstract
ibidi Solutions:
- The ibidi µ-Slides, µ-Dishes, and µ-Plates are ideally suited for transduction using various cell numbers and volumes.
Transduction of Primary Endothelial Cells
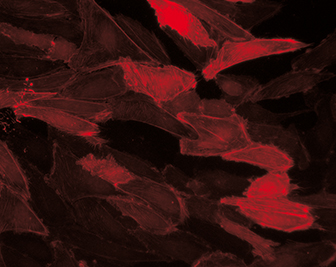
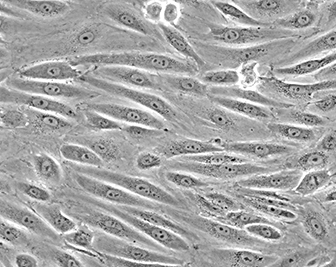
Human umbilical vein endothelial cells (HUVEC) were transduced with an adenoviral LifeAct vector to visualize F-actin and cultured under flow conditions (20 dyn / cm²) in a µ-Slide I 0.4 Luer over 9 days.
Recombinant lentiviral vectors are powerful tools for gene transfer with some advantages over other delivery vectors: Besides cells that undergo mitosis, they also have the ability to transduce non-dividing cells. Further, lentiviruses enable stable gene transfer in vitro and in vivo, as they integrate into the host cell genome and offer the possibility of positive cell selection. They have a broad host cell range that also includes cell types such as primary neurons, lymphocytes, and macrophages. Moreover, lentiviral vectors have also proven to be effective in transducing brain, liver, muscle, and retina in vivo without toxicity or immune responses. Biosafety level S2 is needed for lentiviral transduction.
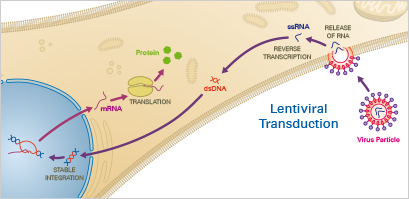
Principle:
Lentiviruses—a subclass of retroviruses—have the ability to permanently integrate into the genome of the host cell. After the virus has entered the cell, the viral RNA is transcribed by the reverse transcriptase to produce double-stranded DNA that enters the nucleus. Finally, the transgene is integrated into the host genome via the lentiviral integrase enzymes. When using lentiviral transduction, the user has to take into account effects caused by genomic integration.
Sakuma T., Barry M.A., Ikeda Y. Lentiviral vectors: basic to translational. Biochem J, 2012, 10.1042/BJ20120146
read abstract
ibidi Solutions:
- The ibidi µ-Slides, µ-Dishes, and µ-Plates are ideally suited for transduction using various cell numbers and volumes.
Read on and learn more about Chemical Transfection or Physical Transfection.




